Density of water at 25 degrees celsius4/13/2024  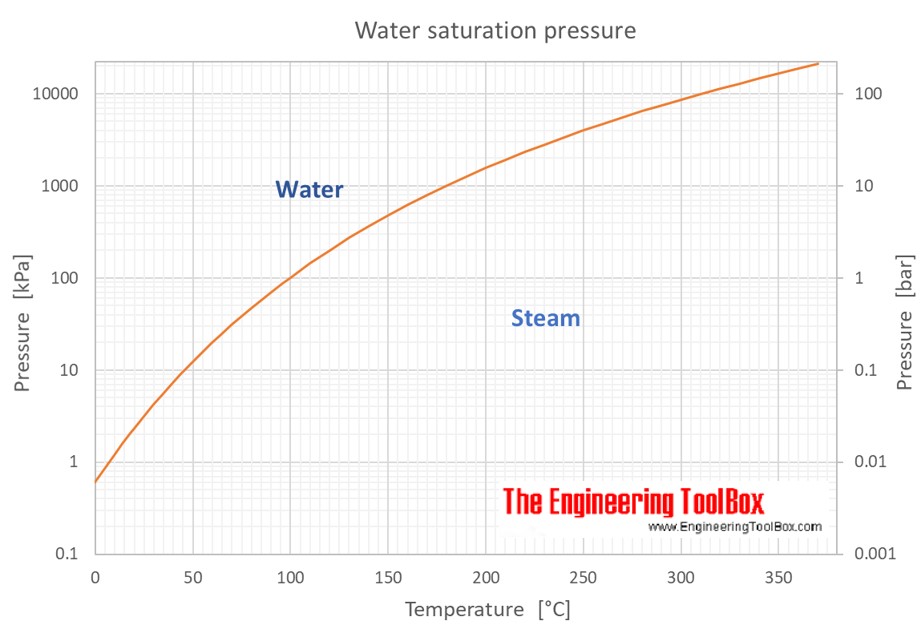
If the temperature of the metal sample rises from 15.0☌ to 40.0☌ as the sample absorbs 89.0 J of heat, what is the specific heat of the sample? Now look at your periodic table and choose a metal that is most likely the identity of the sample. A piece of aluminum with a mass of 100.0 g has a temperature of 20.0☌. How many kilojoules of heat are absorbed when 1.00 L of water is heated from 18☌ to 85☌? (Hint: You first need to determine the mass of the water, then calculate q in the requested unit.)ħ. How much heat is required to raise the temperature of 250.0 g of mercury by 52☌?Ħ. What is the specific heat of the stainless steel?Ĥ. A piece of stainless steel with a mass of 1.55 g absorbs 141 J of heat when its temperature increases by 178☌. What is the specific heat of the olive oil?ģ. When 435 J of heat is added to 3.4 g of olive oil at 21☌, the temperature increases to 85☌. The temperature of a piece of Metal X with a mass of 95.4g increases from 25.0☌ to 48.0☌ as the metal absorbs 849 J of heat. (Specific heat capacity of ethanol is 2.44 J oC -1g -1).ġ. What will be the final temperature of the ethanol if 3240 J was needed to raise the temperature of the ethanol? The initial temperature of 150g of ethanol was 22 oC. (Specific Heat Capacity of aluminum is 0.90 J oC -1g -1).Ĥ. 216 J of energy is required to raise the temperature of aluminum from 15 o to 35 oC. Calculate the specific heat capacity of copper given that 204.75 J of energy raises the temperature of 15g of copper from 25 o to 60 o.ģ. Calculate the amount of heat needed to increase the temperature of 250g of water from 20 oC to 46 oC.Ģ. Reapply to q=mC DT, combine the mass of ice and water, assuming we are at a temp. The ice will melt, so the letft over energy is What is the final temperature after a 21.5 gram piece of ice at 0 is placed into a Styrofoam cup with 125.0 grams of water initially at 76.5 oC? Assume no loss or gain of heat from the surroundings.Įnergy to melt the ice energy to bring the water to 0 oC Solving for Final Temperature when Ice is added to water Specific heat: Quantity of heat necessary. Specific enthalpy: Sensible Heat, it is the quantity of heat contained in 1 kg of water according to the selected temperature. Density: Ratio of the mass of water (kg) occupied in a volume of 1 m3. normal atmospheric pressure on the sea level at 0☌. Solving for the Final Temperature when Metal is dropped in water.ĭetermine the final temperature when a 25.0g piece of iron at 85.0☌ is placed into 75.0grams of water at 20.0☌. Pressure: Atmospheric pressure at 1,01325 bar, i.e. 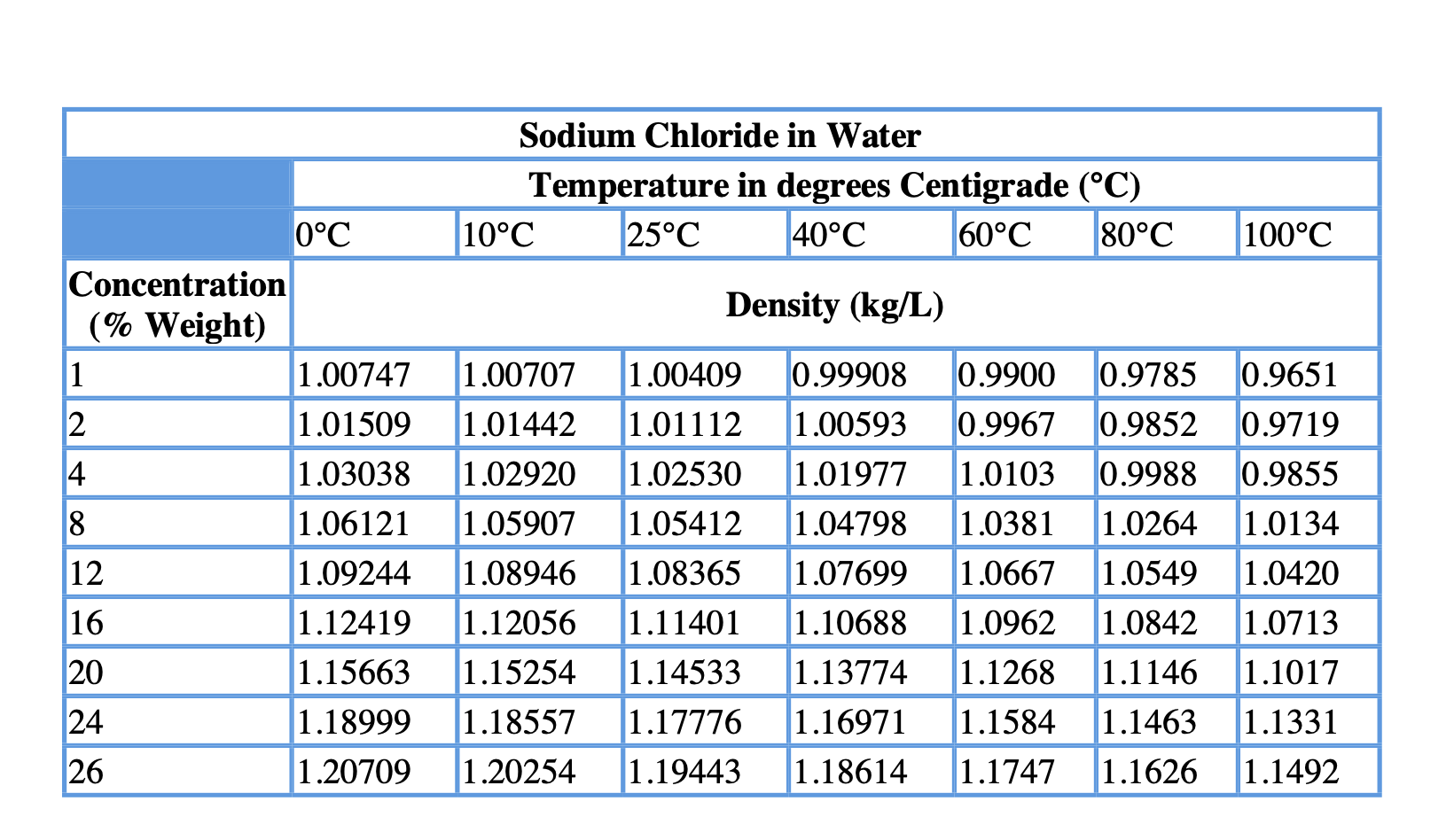
If no heat was lost to the surroundings what is the specific heat of the metal? The final temperature of the water and metal was 34.6 Celsius. A 245.7g sample of metal at 75.2 degrees Celsius was placed in 115.43g water at 22.6 degrees Celsius. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
